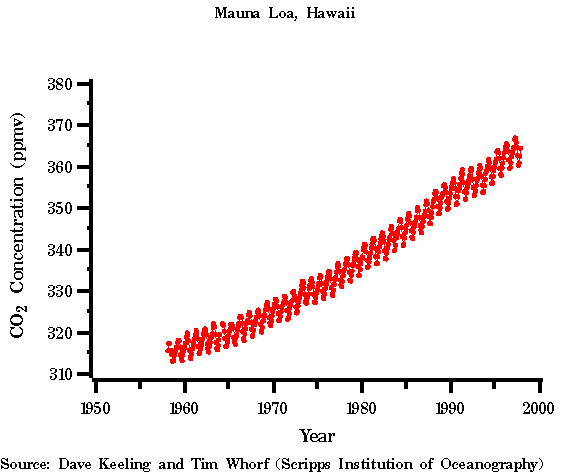
 Figure 1. CO2 measurements from Mauna Loa (from the CDIAC
Web site)
Figure 1. CO2 measurements from Mauna Loa (from the CDIAC
Web site)
The topic of Earth's changing climate has received a lot of political attention over the past decade, especially following the Earth Summit in Rio de Janeiro, Brazil, in 1992. Just about one year ago, at a heavily publicized meeting, the Kyoto Protocol was formulated. This document, which awaits ratification (and will be controversial in the United States this reelection year), calls for reductions in emissions of greenhouse gases, in particular CO2, CH4, N2O, fluorocarbons, and sulfur hexafluoride. In this section of the course, we examine what all the fuss is over.
The Earth's Climate has always been variable. Records of earth's past temperature can be deduced by examining chemicals frozen into glaciers in Greenland and Antarctica. We are living in a relatively warm period in recent history, having just come out of an "ice age" a little more than 10,000 years ago. The spread of modern civilization from the warm Mediterranean region northward through Europe and across land and ice bridges to North America was made possible by the warmer climate that has prevailed for the past 10,000 years. However, there have been recent cold periods, like the Little Ice Age between 1450 and 1850 AD. During this period, the colonists of Greenland were forced to leave.
It is believed that Global Warming will result in a number of changes that will be difficult for humans to adapt to, including loss of property near seaports, increased disease, large numbers of extinctions, and changing weather patterns. Therefore, scientists are very eager to understand the factors that govern climate change.
 Figure 1. CO2 measurements from Mauna Loa (from the CDIAC
Web site)
Figure 1. CO2 measurements from Mauna Loa (from the CDIAC
Web site)
How is it that humans can change the earth's climate? After all, we
are all so small relative to the earth. It is through our collective contributions
that we are changing the concentrations of important molecules in the atmosphere,
like carbon dioxide (CO2), methane (CH4), nitrous oxide (N2O), and fluorocarbons
(FCs). One of the most significant discoveries of the 20th century was
made by Charles
D. Keeling of Scripps Institution of Oceanography. In the 1950s, Professor
Keeling began to measure CO2 in the earth's atmosphere. His measurements,
shown below, showed that mankind's reliance on fossil fuels for energy
were increasing the CO2 in the atmosphere.
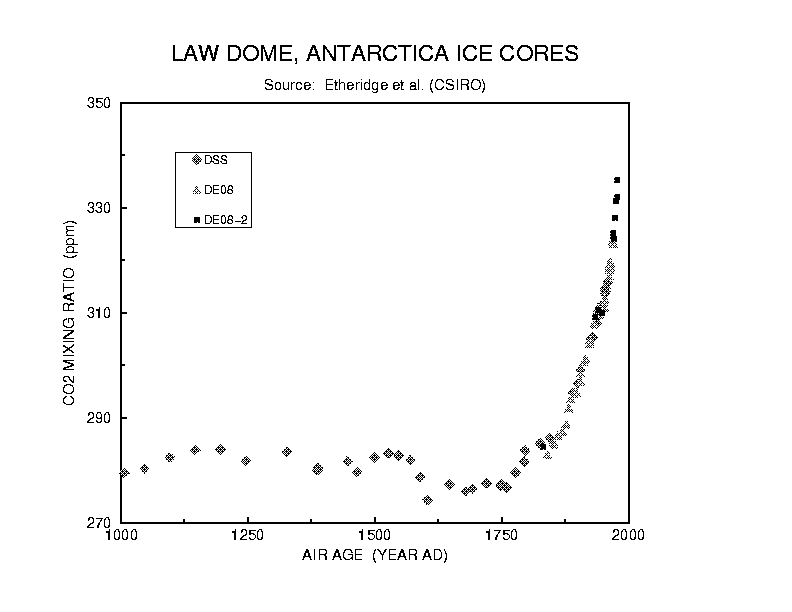 Figure 2. CO2 ice core record (from the CDIAC
Web site )
Figure 2. CO2 ice core record (from the CDIAC
Web site )
Later, by looking at gases dissolved in ice trapped in glaciers, scientists
were able to demonstrate that the abundances of CO2 in the present-day
atmosphere were higher than they had been in nearly 1000 years. Ice cores
from very old antarctic ice show that CO2 fluctuations between 190 parts
per million (ppm) and 270 ppm are correlated with temperature changes of
5-7 degrees Celsius. It is believed that at the present rate CO2 is increasing
in the atmosphere, the average temperature of the globe will increase about
2-4 degree C from present temperature in the next 100 years.
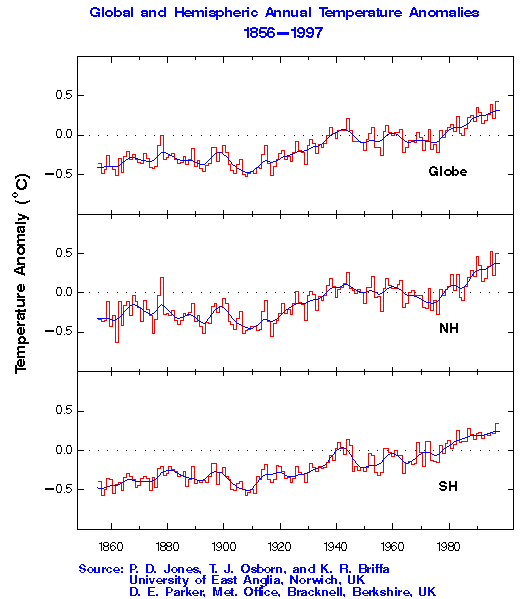 Figure 3. Temperature Trends (from the CDIAC
Web site )
Figure 3. Temperature Trends (from the CDIAC
Web site )
The figure at the left shows a reconstruction of the earth's temperature
in the two hemispheres from 1860 to present. A clear warming trend is present.
However, because natural fluctuations can be of a similar magnitude, it
isn't entirely clear from this record that the cause of the increase is
CO2. There are a number of factors that contribute to global temperatures,
including a "buffering" capacity ("heat capacity") of the ocean, which
acts as a short-term regulator. It may be that some of the effects of increasing
temperatures will not be felt for many years to come. In any case, there
are a number of changes that have already been observed on Earth, some
of which are described in a publication by the EPA (EPA
Global Warming).
CO2 is not the only gas that can affect the radiative balance of the Earth. A few other important gases whose concentrations have been elevated by human activities are summarized here.
Our Changing Planet
summarizes some of the important activities that scientists are carrying
out in order to minimize the uncertainties in predicting future global
change.
Properties of Light
Light is a wave of oscillating electric and magnetic fields that are oriented perpendicular to one another. Light waves travel about 3x10^8 meters/second (we designate the speed of light as the letter "c"). As with other traveling waves, light has both a wavelength ("lambda") and a frequency ("nu"), which are related to each other by the expression
[equation 1] c = lambda x nu
Planck "discovered" that the photon (a smallest "packet" of light) had an energy that was proportional to the frequency (or, equivalently, to the inverse of the wavelength):
[equation 2] E = h x nu
In equation 2, h is the constant (called "Planck's constant) that converts from units of frequency (sec^(-1)) to energy in Joules.
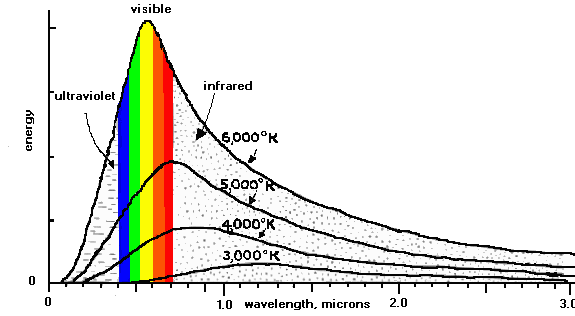
With some calculus, it can be shown that for an object that absorbs and emits light with 100% efficiency (i.e. photons of all colors are absorbed with no reflection and no special colors are emitted) the spectrum ("histogram" of energy emitted versus wavelength) depends simply on the temperature of the object. We call such an "ideal" object a "blackbody", not because its color is black, but because it treats all frequencies equally.
One of the consequences of this relationship between the spectrum of
a blackbody object and its temperature is that the perceived color of the
object changes with temperature. We call such behavior "blackbody
radiation" . The light emitted by a blackbody is a simple function
of temperature.
 (from UCAR
)
(from UCAR
)
The sun is like a blackbody with a temperature of nearly 6000 K. The
earth intercepts some of the light emitted by the sun (but a very tiny
fraction, since we are so far from the sun), converts this light into heat,
and reradiates this energy (in the form of longer wavelength light) back
to space. To estimate the temperature of the earth, we are going to assume
that it, too, radiates like a blackbody. This is true for the surface,
but not for the atmosphere, because there are gases that tend to interact
with specific frequencies in special ways. These gases are called "greenhouse"
gases, and we will deal with them later.
(More on
blackbody radiation)
(More on
temperature)
Earth's Effective Temperature
To estimate the temperature of the earth, we assume that the total amount of energy absorbed is equal to that which is emitted. We call this principle "radiative equilibrium" and the temperature that we calculate the "effective temperature". We can exploit an interesting feature of blackbody radiation, that the energy emitted is proportional to the fourth power of temperature
to write an equation for the temperature of earth, assuming that we
know the amount of solar energy that is absorbed.
T = [So(1-a)/4sigma]^1/4
where So is the solar flux (typically in units of watts/m^2) and a is the albedo (representing the fraction of sunlight that is reflected to space by the earth). Plugging in known values for So (1370 w/m^2) and a (0.3) we calculate that the earth's effective temperature (that average temperature at which the earth must radiate to space to stay in balance with the solar energy absorbed) is about 255 K. This is far too low to support liquid water (freezing point of 273 K), so what accounts for the fact that the earth's surface is much warmer?
We need to consider the effect of the atmosphere on the balance of the radiation at the surface. As shown in class, if we put an absorbing layer completely around the earth at, say, 5 km altitude, we find that this layer must achieve an average temperature of 255 K in order to satisfy radiative equilibrium. However, this layer will radiate both upward to space and back to the surface. It is this additional energy input, sigmaT^4 (where T= 255 K) that gets added to the energy from the sun in order to determine the new balance for the surface. Assuming the evenly distributed atmosphere acts like a single sheet of absorbing material, we find that the surface temperature will warm from 255 K (without the layer) to 303 K (86 F), plenty warm for liquid water!
In reality, the atmosphere doesn't absorb all frequencies equally well. Some are transmitted directly to space, while others are absorbed more than once between their trip from the surface to space. It is in the remaining "windows" in the emission spectrum that gases such as CFCs, N2O, and CH4 trap additional radiation that doesn't get trapped by CO2 and water vapor, the two most important greenhouse gases. The textbook has a pretty good description of the greenhouse effect. If you would like additional information, try the NOVA Greenhouse Page